Bio-Share | Patient-Friendly Injection Solutions: Prefilled Syringes, Cartridges & Pens
Addtime: Aug 25 , 2025
Amid the growing trend of biologics moving toward home-based and portable use, the combination of prefilled syringes, cartridges, and pen injectors is rapidly gaining market share. From insulin to immunotherapies, these formats are becoming the global standard for injectable products, reshaping the way medicines are delivered.
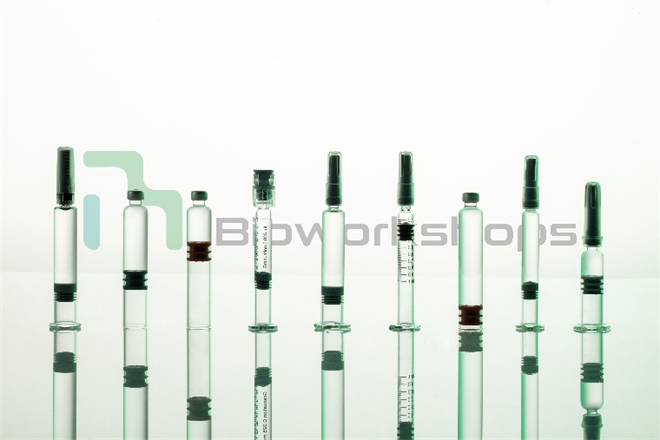
Figure 1: Bioworkshops provides aseptic filling for “combination products” such as prefilled syringes, cartridges, and pen injectors.
Taking cartridges as an example: as the core liquid storage unit of pen injectors, cartridges are typically composed of a highly inert glass barrel [1], a plunger that drives drug delivery, and sealing components that prevent contamination. Some designs also incorporate a glass ball [2,3], allowing the suspension to be evenly dispersed before use through a simple inversion, thereby ensuring accurate dosing and consistent efficacy. These structures are not mere mechanical assemblies; rather, they are carefully engineered to maintain drug stability during storage and administration, prevent contamination, and ensure precise dosing with every actuation.
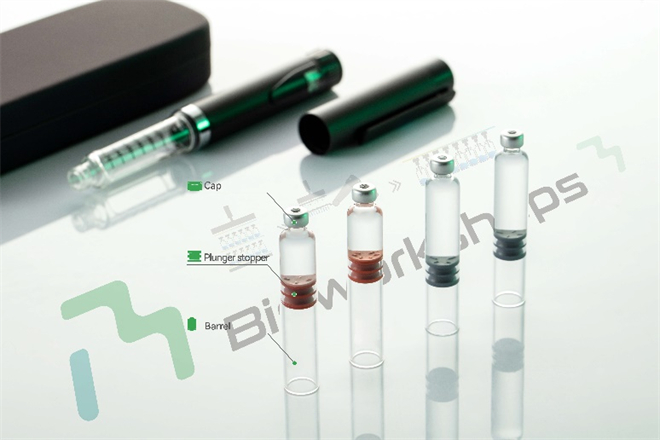
Figure 2: Cartridge–pen injector “combination product.”
With a stable liquid storage system in place, pen injectors offer a patient-friendly mode of administration—requiring only a simple twist or press to complete the injection. The design is easy to use, convenient to carry, and helps reduce discomfort during drug delivery. Especially in chronic disease management, the combination of replaceable prefilled syringes/cartridges and adjustable dosing enables patients to carry out treatment confidently in daily life, enhancing their ability to manage therapy at home.
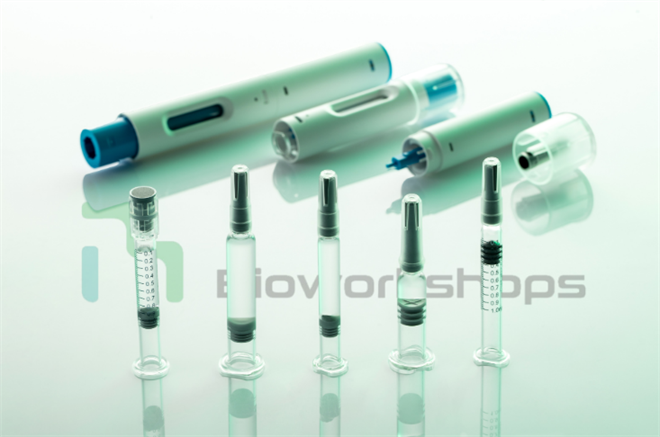
Figure 3: Prefilled syringe–pen injector “combination product.”
From a regulatory perspective, the combination of prefilled syringes, cartridges, and pen injectors is generally classified as a “combination product.” Such products must meet both the quality and safety requirements of pharmaceuticals as well as the performance and regulatory standards of medical devices. For example, the U.S. FDA, in its guidance Technical Considerations for Pen, Jet, and Related Injectors [4], explicitly requires that aseptic filling comply with GMP, that products undergo safety and performance verification, and that packaging, labeling, and instructions for use be subject to review. Regulatory requirements therefore cover all critical stages of the product’s lifecycle:
Aseptic Assurance: Filling and sealing operations must be performed in a controlled environment compliant with GMP requirements, ensuring sustained and effective sterility.
Performance Verification: Devices must undergo comprehensive testing—including dose accuracy, repeatability, seal integrity, leakage prevention, and contamination resistance—and demonstrate stable performance throughout the entire lifecycle of use.
Human Factors and Usability Evaluation: The injector design must be validated through Human Factors Engineering (HFE), ensuring intuitive operation in real-world conditions with minimized risk of misuse.
Materials and Compatibility: All components in direct contact with the drug product (e.g., glass barrel, plunger, sealing elements) must undergo compatibility and chemical stability assessments to prevent leachables, extractables, or adsorption that could compromise drug efficacy.
Labeling and Instructions for Use: Packaging, labeling, and instructions must be reviewed to ensure clear communication of handling methods, storage conditions, shelf life, and safety precautions, avoiding ambiguity or misinterpretation.
Change Control and Traceability: Any changes to design, materials, or processes must undergo re-evaluation and verification, with traceable records maintained within the quality management system.
This places clear requirements on production line configuration and processes: an aseptic barrier environment (isolator / oRABS) to minimize contamination risks; high-precision filling systems to ensure dose consistency; reliable detection of visual defects and particulates; and the capability for rapid changeover between different formats and batch sizes. At the same time, manufacturing must operate under standardized process validation and continuous monitoring to ensure consistency and full traceability.

Figure 4: Independent robotic arms on Bioworkshops’ aseptic filling line
Bioworkshops’ cartridge aseptic filling line is equipped with isolator systems and independent robotic arms, minimizing contamination risks from human intervention. The system accommodates multiple cartridge formats and supports both small-batch clinical supply and large-scale commercial production. All critical quality attributes and in-process data are fully traceable, while rapid mold changeovers enable shorter batch-to-batch transitions and enhance multi-product manufacturing efficiency.
With the growing adoption of cartridges and pen injectors, Bioworkshops provides stable and compliant aseptic filling support across different project stages.
Prefilled Syringe / Cartridge Aseptic Filling Line Capabilities
- Filling volume range: 0.2 mL – 5.0 mL
- Filling speed: up to 12,000 units per hour
- Robotic aseptic prefilled system: ensures high-precision dosing and robust sterility assurance
Reference
[1] Stevanato Group. PDA Training: Introduction to Cartridge Systems and Components. 2025.
[2] Novo Nordisk. NovoPen® 4 Product Information (UK). 2024.
[3] Frontiers in Endocrinology. Technological Advances in Pen Injectors for Biopharmaceutical Delivery. 2022.
[4] U.S. Food and Drug Administration (FDA). Technical Considerations for Pen, Jet, and Related Injectors Intended for Use with Drugs and Biological Products. 2023.
News
-
2025-08-07
Platform Strategies for Complex Antibody Expression: From Vector Engineering to System-Level Optimization
More -
2025-08-25
Bio-Share | Patient-Friendly Injection Solutions: Prefilled Syringes, Cartridges & Pens
More -
2024-01-18
Bioworkshops has been selected as a "Unicorn" incubator in Suzhou in 2023
More






