Platform Strategies for Complex Antibody Expression: From Vector Engineering to System-Level Optimization
Addtime: Aug 07 , 2025
As of July 2025, a total of 20 bispecific antibodies (#BsAbs) have been approved worldwide (Figure 1). The majority are indicated for cancer immunotherapy, while a smaller number target chronic inflammatory and hematological diseases. Currently, more than 200 bispecific antibodies are in clinical development for applications such as radioimmunotherapy, antiviral therapies (e.g., Ebola), neurological diseases (e.g., Alzheimer’s disease), and solid tumor treatments [1,2,3].
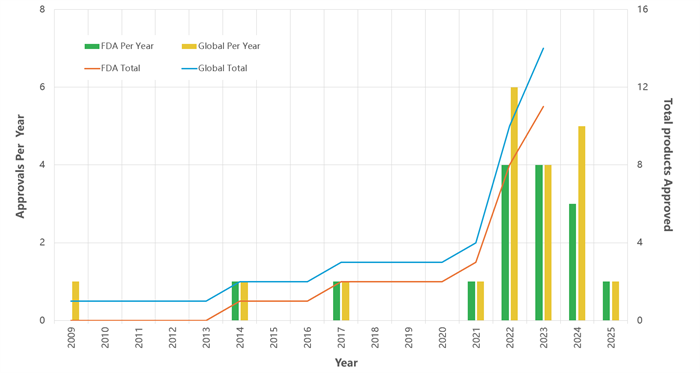
Figure 1. Global approvals of bispecific antibodies (2009–2025)
Bispecific antibodies exert their therapeutic effects through a variety of mechanisms of action (MoAs), including: cell bridging (e.g., mosunetuzumab-axgb), coagulation factor bridging (e.g., emicizumab-kxwh), receptor activation or blockade (e.g., ozoralizumab and faricimab-svoa), receptor or ligand internalization/aggregation, cofactor mimicry (e.g., emicizumab-kxwh), and redirection of cytotoxic effector cells.
The diversity of mechanisms of action and therapeutic target requirements has led to a wide range of BsAb molecular architectures. This structural heterogeneity directly contributes to their complex physicochemical profiles, including variations in molecular weight, isoelectric point, charge heterogeneity, and aggregation propensity.
The key physicochemical properties of bispecific antibodies include protein sequence and molecular size, spatial conformation (secondary/tertiary structure), hydrophobicity, charge distribution, isoelectric point, post-translational modifications (e.g., glycosylation, deamidation, oxidation), and molecular structure (e.g., symmetry and aggregation state). To date, more than 100 BsAb structural formats and over 30 enabling technology platforms have been reported. The selection of a specific format (e.g., IgG-based or antibody fragments) is primarily driven by therapeutic application, MoA, functional requirements, and developability.
The structural, physicochemical, and functional diversity of BsAbs necessitates tailored process development strategies to ensure product quality, stability, and scalability. Although rational molecular design can reduce issues such as heavy- and light-chain mispairing, it cannot fully eliminate them. Consequently, cell line development serves as a critical starting point in process development, enabling efficient protein expression, accurate chain assembly, and reliable scalability.
In antibody expression, particularly for structurally complex molecules such as BsAbs, achieving coordinated expression of heavy and light chains, increasing yield, and ensuring proper folding are critical factors for upstream development efficiency. Studies have shown that optimizing vector architecture, especially promoter design, can significantly improve antibody yield and quality. For example, a study [4] employing a dual-promoter single-plasmid system for full-length IgG demonstrated that placing two enhanced CMV (eCMV) promoters symmetrically within the same plasmid enabled preferential light-chain expression, increased correct assembly rates, and markedly enhanced protein yields in both Expi293-F and ExpiCHO-S cells. Compared with the conventional dual-plasmid system, this strategy offers advantages in expression efficiency, operational simplicity, and product consistency, providing a valuable approach for the expression optimization of complex antibodies such as BsAbs.
To achieve synchronized expression of heavy and light chains, researchers developed a modular vector incorporating a bidirectional promoter (BiDi). As shown in Figure 2C, after Golden Gate cloning, the heavy- and light-chain genes were inserted into the same plasmid and co-expressed under the control of two symmetrically arranged eCMV promoters. Figure 2D illustrates the structural schematic of an IgG1 antibody.
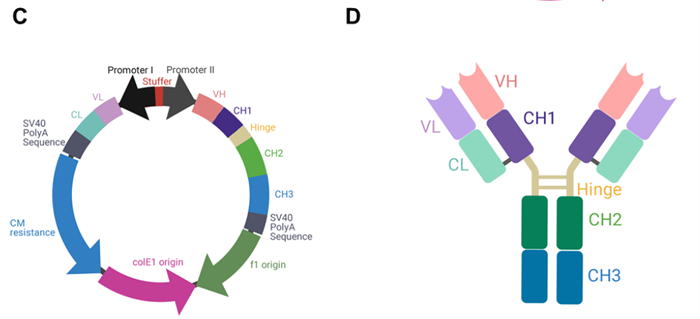
Figure 2. Schematic diagram of the dual-promoter single-plasmid system and its expression products
As shown in Figure 3, the optimized 2xeCMV promoter system achieved the highest antibody expression levels in Expi293-F cells, demonstrating a significant improvement over conventional designs. This result clearly illustrates that rational tuning of vector architecture can effectively enhance antibody expression efficiency.
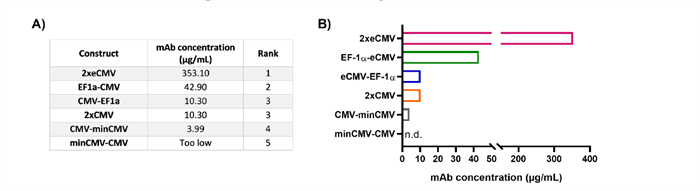
Figure 3. Schematic of a dual-promoter single-plasmid system and its antibody expression products
To evaluate system universality, the study further compared the performance of the 2xeCMV system and the traditional dual-plasmid system in expressing κ-type antibody Durvalumab and λ-type antibody Avelumab. As shown in Figure 3, both antibodies exhibited comparable expression levels across systems, indicating that the single-plasmid system is independent of light-chain type, highly adaptable, and suitable for multiple antibody formats, including bispecifics.
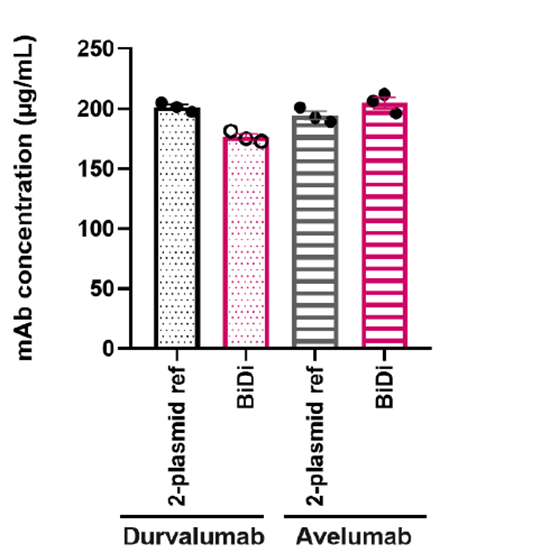
Figure 4. Effects of different dual-promoter combinations on antibody expression levels
The above findings were obtained from single-plasmid experiments on conventional monoclonal antibodies. Beyond dual-promoter single-plasmid designs, some studies [5] have explored optimization of plasmid input ratios during transfection to improve transfection efficiency and overall antibody yield for bispecific constructs.
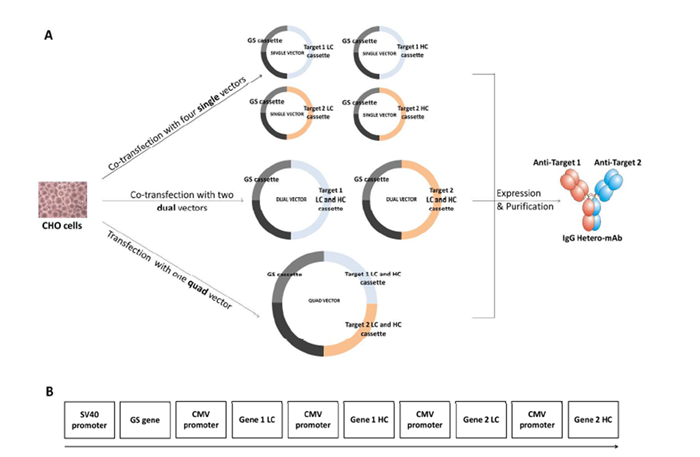
Figure 5. Validation of the dual-promoter single-plasmid system across antibodies with different light chain types
To coordinate the expression of the four chains of bispecific antibodies (BsAbs), the study compared three plasmid construction strategies (Figure 5A). Traditionally, a four-plasmid system is used, where each heavy and light chain is encoded on a separate plasmid; however, this approach is complex and difficult to balance in terms of expression ratios. To simplify, the team developed a dual-vector system, with each plasmid encoding one HC-LC pair, and further advanced a single-plasmid system (Quad vector), which integrates all four chains into one construct (Figure 5B).
In the single-plasmid system design, each chain is driven by an independent promoter and signal peptide sequence, with a GS selection marker integrated into the construct. This design allows the plasmid to be used not only for transient expression but also for generating stable CHO cell lines. It streamlines transfection operations and avoids chain mispairing issues caused by imbalanced plasmid ratios in multi-vector systems.
The study compared the performance of dual-vector and quad-vector systems for expressing IgG-type BsAbs in CHO cells. Results showed that while both systems exhibited similar antibody expression levels and assembly efficiency in transient expression, the quad-vector design demonstrated higher assembly accuracy, lower mispaired product ratios, and more stable and controlled expression in stable CHO systems.
Therefore, whether using single- or dual-vector systems, expression and screening strategies can be tailored to the physicochemical characteristics of each molecule, supported by transient expression assays and high-throughput screening methods. Incorporating process development considerations early in the molecular design stage enables feasibility assessments of expression and manufacturability from the outset, while also aligning downstream scale-up and quality control requirements. This integrated approach facilitates co-optimization of molecular design and process development, ultimately improving overall efficiency and success rates.
Vector optimization has markedly improved the developability of BsAbs, while engineering of host cells has provided additional opportunities. To investigate the impact of chain stoichiometry on Fab–scFv–Fc production, researchers designed a polycistronic vector based on internal ribosome entry sites (IRES), enabling differential expression of three polypeptide chains through vector design [6]. The highest yield of correctly assembled products was achieved when the heavy chain (HC) and scFv–Fc chain were expressed at a 1:1 ratio, with an excess of the light chain (LC). Compared with IRES- and multi-promoter–driven co-expression, use of 2A peptides to co-express the three chains resulted in the highest product titer and the greatest proportion of correctly assembled molecules. Recombinase-mediated site-specific integration systems can further enable precise regulation of chain ratios.
BsAbs face three major challenges during preclinical development. First, as non-naturally occurring molecules, their expression levels can be unpredictable, thereby affecting developability. Second, their complex structures generate diverse impurities, and certain mispaired species share physicochemical properties with correctly assembled molecules, creating significant downstream purification challenges. Third, during cell line construction, the ratio of transfected chain-encoding plasmids strongly influences mispair formation, and different BsAb formats often require distinct vector system choices.
To address these process challenges, we have progressively established a platform-based development system tailored to the structural and expression complexities of bispecific, trispecific, and other multispecific antibodies. This framework encompasses key stages such as cell line construction, cell culture, purification process development, and analytical method development. By employing transient expression tests to predefine chain stoichiometry, we are able to selectively identify minipools and monoclonal cell lines with reduced impurity formation and lower proportions of hard-to-remove mispaired products during stable cell line development. This system, validated and applied across multiple projects, enables flexible strategy adjustments for different antibody architectures and functional mechanisms, thereby achieving higher expression yields and enhanced process robustness.
Reference:
1. Ma J, et al. Bispecific Antibodies: From Research to Clinical Application. Front. Immunol. 12, 2021: 1–19; https://doi.org/10.3389/fimmu.2021.626616.
2. Goebeler M-E, Stuhler G, Bargou R.Bispecific and Multispecific Antibodies in Oncology: Opportunities and Challenges. Nat.Rev. Clin. Oncol. 21(7) 2024: 539–560; https://doi.org/10.1038/s41571-024-00905-y.
3. Bratt J, et al. Therapeutic IgG-Like Bispecific Antibodies: Modular Versatility and Manufacturing Challenges, Part 1.BioProcess Int. 15(11) 2017: 36–42; https://www.bioprocessintl.com/manufacturing/therapeutic-igg-like-bispecific-antibodiesmodular-versatility-and-manufacturingchallenges-part-1.
4. Stefania C. Carrara. et al. Recombinant Antibody Production Using a Dual-Promoter Single Plasmid System. Antibodies 2021, 10, 18. https://doi.org/10.3390/antib10020018;
5. Yashas Rajendra et al. Transient and Stable CHO Expression, Purification and Characterization of Novel Heterodimeric Bispecific IgG Antibodies. Biotechnology Progress. DOI 10.1002/btpr.2414
6. Ong HK, Nguyen NTB, Bi J, Yang Y. Vector design for enhancing expression level and assembly of knob-into-hole based FabscFv-Fc bispecific antibodies in CHO cells. Antib Ther. 2022 Oct 13;5(4):288-300. doi: 10.1093/abt/tbac025. PMID: 36518226; PMCID: PMC9743168.
Bioworkshops Complex Antibody Platform
Bioworkshops has established a robust multi-specific antibody development platform, dedicated to the full lifecycle of complex antibody programs, including bispecifics and trispecifics:
Diverse Molecular Design Support — Covering symmetric, asymmetric, and IgG-like formats, with early-stage design assessment and technical guidance to accelerate downstream development.
High-Expression Systems — Optimized plasmid construction and transfection strategies, combined with high-yield stable cell line development, to significantly enhance antibody expression and correct assembly rates.
End-to-End Development Framework — From molecular design assessment, cell line development, and process optimization to GMP manufacturing, ensuring product quality and scalability.
Proven Project Experience — Successfully delivered multiple clinical-stage BsAb projects across oncology, inflammation, and autoimmune diseases.
News
-
2025-08-07
Platform Strategies for Complex Antibody Expression: From Vector Engineering to System-Level Optimization
More -
2025-08-25
Bio-Share | Patient-Friendly Injection Solutions: Prefilled Syringes, Cartridges & Pens
More -
2024-01-18
Bioworkshops has been selected as a "Unicorn" incubator in Suzhou in 2023
More






