Bio Share | TsAb: Innovations and Challenges of Next-Generation Antibody Therapies
Addtime: Mar 21 , 2025
At present, 18 bispecific antibodies (BsAbs) have been approved worldwide, with the majority indicated for oncology. Among them, CD20×CD3 accounts for the largest share (4 products). Meanwhile, with continuous advancements in antibody engineering technologies and deeper understanding of mechanisms of action, trispecific antibodies (TsAbs)—which can simultaneously target three distinct antigen epitopes on tumor cells or immune effector cells—have emerged as an important breakthrough in oncology therapeutics. TsAbs and multispecific antibodies (MsAbs) are becoming strong contenders for the next generation of antibody therapies.
Advantages and Strategic Landscape of TsAbs
Compared with traditional monoclonal and bispecific antibodies, trispecific antibodies can simultaneously target three different antigens or receptors, offering more precise and effective therapeutic strategies. They hold enormous potential particularly in oncology immunotherapy, cell therapy, and autoimmune diseases. Currently, leading global biopharmaceutical companies including Sanofi, Amgen, Roche, Genentech, Johnson & Johnson, and Pfizer are actively developing TsAbs, with several candidates already in clinical trials.
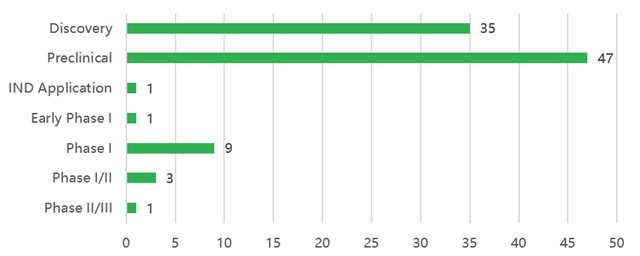
Figure 1. Global progress in trispecific antibody development
Clinical Design Strategies for TsAbs
At present, about 39 trispecific antibodies have entered clinical development. The major design strategies include:
- T Cell Engagers (TCEs): Enhance T cell activation and promote T cell recruitment at tumor sites.
- NK Cell Engagers (NKCEs): Enhance NK cell–mediated antibody-dependent cellular cytotoxicity (ADCC).
- Immune Checkpoint Blockade: Relieve immunosuppression in the tumor microenvironment to improve immune efficacy.
- Targeting Multiple Tumor-Associated Antigens (TAAs): Improve antibody specificity and cytotoxic effects.
- Human Serum Albumin (HSA) Binding Strategy: Prolong serum half-life and enhance drug stability via albumin binding, reducing dosing frequency.
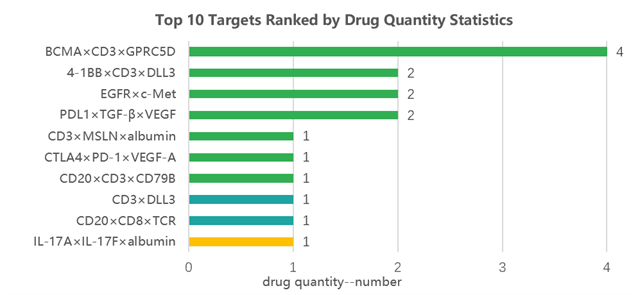 Figure 2. Summary of targets of trispecific antibodies in clinical research
Figure 2. Summary of targets of trispecific antibodies in clinical research
Manufacturing Challenges and Solutions for TsAbs
Due to their structural complexity, stringent production requirements, and stability challenges, trispecific antibodies (TsAbs) face numerous technical difficulties during process development.
First, cell line construction is the critical starting point for TsAb development. Owing to their intricate structure and unique design, tailored strategies are required. Since cell line development is typically time-consuming, well-designed preliminary studies can greatly reduce downstream risks and difficulties, thereby avoiding project delays.
In practice, TsAbs can be divided into symmetric and asymmetric formats. Symmetric TsAbs often have larger molecular weights, which increases the expression burden for cells and thus requires particular attention to expression risks. For asymmetric TsAbs, the major challenge lies in homodimer impurities. Designing experiments at the cell line stage to minimize the proportion of homodimers is critical for reducing future purification complexity. To this end, we frequently conduct vector ratio testing to assess the expression levels of different chains within cells, balance chain expression, and thereby reduce homodimer formation caused by expression imbalance.
During this optimization process, analytical method development must be introduced early to identify homodimers. Moreover, the principles behind these analytical methods can provide guidance for downstream purification development.
Beyond homodimers, purification process optimization for TsAbs faces additional challenges: these molecules may contain scFv fragments, Fab fragments, VHH fragments, or aggregates. Conventional chromatography platforms are not always applicable; thus, broader resin screening is necessary, together with careful monitoring of intermediate stability.
To address these challenges, in our actual project deliveries we have established platformized strategies for cell line development, purification process development, and analytical method development tailored to trispecific and other multispecific antibodies. These strategies have been successfully applied to multiple projects, ensuring that antibody products maintain outstanding quality control and stable manufacturability throughout industrialization.
News
-
2025-08-07
Platform Strategies for Complex Antibody Expression: From Vector Engineering to System-Level Optimization
More -
2025-08-25
Bio-Share | Patient-Friendly Injection Solutions: Prefilled Syringes, Cartridges & Pens
More -
2024-01-18
Bioworkshops has been selected as a "Unicorn" incubator in Suzhou in 2023
More






