Towards Comprehensive Characterization of Antibody Drugs: Focus on Physicochemical Properties
Addtime: Dec 05 , 2023
The monoclonal antibody (mAb) technology was invented in 1976 by Georges Köhler and César Milstein. Since then, it has undergone four major stages of development: murine, chimeric, humanized, and fully human antibodies. Because murine and chimeric antibodies can trigger human anti-mouse antibody (HAMA) responses, humanized and fully human antibodies have become the mainstream focus of biotechnology companies. Monoclonal antibodies have a molecular weight of approximately 150 kDa and possess a highly complex structure, making their quality control requirements far more stringent compared with small-molecule drugs.

Based on monoclonal antibody technology, various emerging modalities have subsequently been developed, including antibody-drug conjugates (ADCs), multispecific antibodies, and nanobodies. A major challenge in the development of antibody therapeutics is the comprehensive characterization of their critical quality attributes (CQAs). This article series is divided into two parts: the first (Part I) focuses on physicochemical properties, including antibody structure, charge and size variants, glycosylation heterogeneity, process-related impurities, and aggregate analysis; the second (Part II) will cover activity analyses, including binding activity and biological activity.
Physicochemical Characterization
1. Antibody Structure
Structural characterization of antibody therapeutics primarily includes the following parameters:
(1)intact molecular weight,
(2)light chain molecular weight,
(3)heavy chain molecular weight,
(4)amino acid sequence,
(5)disulfide bonds,
(6)free thiols,
(7)glycosylation sites,
(8)N-terminal sequencing,
(9)post-translational modifications (PTMs), and
(10)secondary and tertiary structure characterization.
Among the first nine items, LC/MS is the most essential and fundamental analytical method. Secondary and tertiary structures are typically examined using circular dichroism (CD) and infrared spectroscopy, details of which are not elaborated here.
The purpose of antibody structure characterization, beyond achieving a comprehensive understanding of product quality, is to identify critical amino acid residues and post-translational modifications (PTMs) that could impact therapeutic efficacy. Such insights enable targeted process modifications and confirmation, thereby avoiding adverse effects during late-stage process development or clinical phases. This article will primarily discuss several key modifications: deamidation, isomerization, oxidation of Met and Trp residues, and unpaired cysteines.
1.1 Deamidation
Asparagine (Asn) deamidation—particularly of residues located in the complementarity-determining regions (CDRs)—is one of the most common degradation pathways for monoclonal antibody drugs. When an Asn residue is followed by a small and reactive glycine (Gly) residue (NG motif), it is especially prone to deamidation. If such events occur in the CDR regions, they can reduce antigen-binding affinity and result in loss of potency. Therefore, during antibody sequencing and bioinformatic assessment, special attention should be paid to Asn-Gly residues in the CDRs, and their stability should be evaluated under forced degradation conditions.
Additionally, the IgG Fc region contains the “PENNY” peptide loop, which also harbors deamidation-prone sites. However, since these generally do not negatively affect antigen binding, they are usually not prioritized for monitoring.
1.2 Oxidation of methionine
Methionine (Met) and tryptophan (Trp) residues are highly susceptible to oxidation. Oxidation of methionine residues in the heavy-chain CDR2 region or in the Fc domain typically does not affect antigen binding. However, when oxidation levels are elevated, or when critical CDR residues are affected, antigen-binding affinity and overall therapeutic efficacy can be significantly reduced.
Met residues located near the CH2–CH3 interface are particularly prone to oxidation, which can decrease thermal stability, increase aggregation, reduce complement-dependent cytotoxicity (CDC), lower binding affinity to FcRn, and shorten the in vivo half-life.
Similarly, oxidation of Trp residues in CDRs may compromise efficacy, decrease thermal stability, and increase aggregation propensity.
Therefore, during bioinformatic analysis and primary sequence determination, special attention should be paid to Met and Trp residues in the CDRs to assess their susceptibility, and, where necessary, implement control strategies during process development.
1.3 Aspartic Acid Isomerization
Aspartic acid (Asp) isomerization is another degradation pathway in monoclonal antibody drugs. Asp residues located in the CDR regions are particularly prone to isomerization when followed by Gly, His, or Ser residues. Such isomerization in the CDRs can reduce antigen-binding affinity. The process is favored under acidic conditions around pH 5. Therefore, in antibody formulation development, it is important to avoid buffer conditions near pH 5 whenever possible.
1.4 Glycosylation Sites
The IgG CH2 domain contains an N-linked glycan at Asn-297, typically represented by a core heptasaccharide composed of four N-acetylglucosamine (GlcNAc) residues and three mannose residues. Depending on the functional requirements of the antibody, glycoengineering strategies can be employed to alter glycan patterns, thereby modulating functional properties such as effector function or serum half-life. Consequently, glycosylation site detection and analysis are integral throughout the antibody development process and are considered critical quality attributes for product release.
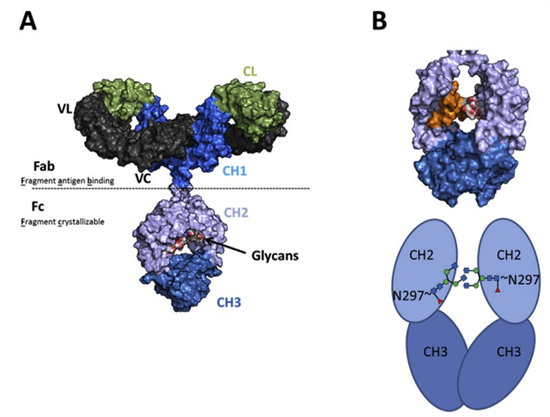
▲Biologicals (2017)
Major glycoengineering strategies include fucosylation, GlcNAc modification, galactosylation, and sialylation. Specifically:
- Fucosylation is catalyzed by core fucosyltransferase (Fut8), adding fucose to the innermost GlcNAc;
- GlcNAc branching is introduced by N-acetylglucosaminyltransferase III (GNT3) at mannose branches;
- Galactosylation is mediated by β-1,4-galactosyltransferase 1 (b4GALT1), adding galactose to terminal GlcNAc residues. These three modifications primarily alter binding affinity to FcγR type I receptors.
Furthermore, by introducing α2,6-sialyltransferase (ST6GAL1), sialylation can be enhanced, thereby improving binding to FcγR type II receptors.
Analytical methods for glycan heterogeneity include LC/MS for glycan identification, HILIC-FLD for quantitative profiling, and boronate affinity chromatography for glycation detection.
1.5 Unpaired Cysteine (Cys) Residues
Unpaired cysteine residues can increase antibody heterogeneity and may significantly affect chemical and thermal stability, biological function, aggregate formation, antigen-binding potency, and protein folding. Although relatively rare, monoclonal antibodies may contain unpaired cysteine residues in their CDR regions. These unpaired cysteines are prone to modification by free cysteine present in cell culture media, which can reduce antigen-binding affinity. Therefore, unpaired cysteine residues should be routinely monitored and characterized as a quality attribute to ensure structural integrity and overall product quality.
2. Variant Analysis
During cell culture, purification, and formulation, recombinant antibodies are prone to developing size and charge variants. These variants are key indicators for antibody identity and consistency assessment.
Common methods for detecting size variants include size-exclusion chromatography (SEC) and CE-SDS (included in the 2015 Pharmacopoeia and revised in the 2020 edition).
Charge heterogeneity in antibody therapeutics reflects the combined outcome of multiple post-translational modifications and is highly sensitive to process changes. Therefore, close monitoring throughout development is essential to ensure consistency. Common characterization techniques include ion-exchange chromatography and isoelectric focusing (IEF, also included in the 2020 Pharmacopoeia), with critical parameters such as isoelectric point (pI) and acidic/basic variants.
3. Aggregate Analysis
Antibody molecules can undergo conformational changes due to post-translational modifications, shear stress during handling, impurities, or pH fluctuations, which may lead to complementary interactions (structural or electrostatic) and the formation of aggregates. Aggregates (or particulate matter) are the most common product-related impurities. Antibody aggregation increases immunogenicity, promotes anti-drug antibody (ADA) formation, and reduces therapeutic efficacy, making close monitoring essential.
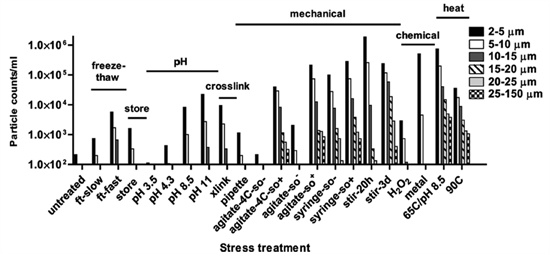
▲THE JOURNAL OF BIOLOGICAL CHEMISTRY 2011
Common analytical methods include SDS-PAGE or CE-SDS, which can detect monomers, fragments, and covalent aggregates under reduced or non-reduced conditions. Size-exclusion chromatography (SEC) is the most widely used method for detecting high-molecular-weight aggregates such as dimers, trimers, or oligomers.

For larger aggregates (particulates), light scattering techniques can be employed to detect particles ranging from <1 nm to 1–10 μm, such as dynamic light scattering (DLS). For subvisible particles, compendial methods include light obscuration (e.g., HIAC) and microscopic counting. Microfluidic imaging is used to analyze particles in the 2–100 μm range and can differentiate proteinaceous aggregates from other extrinsic particles to some extent.
4. Process-Related Impurities
Process-related impurities are those introduced during manufacturing, primarily including host cell DNA, host cell proteins (HCPs), and residual protein A from purification. While host cell proteins generally do not cause severe adverse reactions such as allergies, they serve as excellent quality control indicators—particularly for purification—and are therefore considered critical quality parameters in antibody characterization.
For host cell DNA, quantitative PCR (qPCR) is the most commonly used method. For HCPs, ELISA is generally recommended, although its coverage may be incomplete; therefore, mass spectrometry or targeted Western blotting against specific proteins is often used in combination to ensure comprehensive detection. Residual protein A is typically monitored using ELISA.
5. General Attributes
Beyond the aforementioned critical quality attributes, other general physicochemical parameters are also important for antibody characterization. These include pH, osmolality, appearance, and viscosity, which can be measured following standard methods outlined in pharmacopeial guidelines.
Summary
Compared with small-molecule drugs, antibody therapeutics are several hundred times larger in molecular weight and are more prone to post-translational modifications, size variants, charge variants, and aggregation during expression and processing. These factors pose significant challenges for quality characterization. This article has provided a brief overview of selected parameters that critically influence product quality and, in particular, therapeutic efficacy.

Analytical Laboratory at Bioworkshops
Bioworkshops’ analytical laboratory is equipped with state-of-the-art instruments, including the Thermo Orbitrap Exactive HF-X LC-MS system, Thermo Vanquish UHPLC, ProteinSimple Maurice, SCIEX PA800 plus, Waters ACQUITY H-Class UPLC, Thermo CAD detector, Malvern automated capillary differential scanning calorimeter, Octet system for biomolecular interaction analysis, Molecular Devices M5 microplate reader, and Roche480 qPCR system. With these advanced technologies, the laboratory supports method development, qualification, and validation for monoclonal antibodies, bispecifics, fusion proteins, ADCs, and other biologics. Comprehensive protein characterization studies can also be performed, including process comparability assessments and biosimilarity evaluations. Bioworkshops is committed to providing high-quality, reliable analytical services to support antibody drug development.
Reference:
1. Xiaojie Yu et al,Improving Antibody-Based Cancer Therapeutics Through Glycan Engineering,BioDrugs DOI 10.1007/s40259-017-0223-8
2. Yingda Xu et al,Structure, heterogeneity and developability assessment of therapeutic antibodies,mAbs,2018
3. Cymer, F., Therapeutic monoclonal antibody N-glycosylation – Structure, function and therapeutic potential, Biologicals (2017),
4. Classification and Characterization of Therapeutic Antibody Aggregates ,THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 286, NO. 28, pp. 25118–25133, July 15, 2011.
News
-
2025-08-07
Platform Strategies for Complex Antibody Expression: From Vector Engineering to System-Level Optimization
More -
2025-08-25
Bio-Share | Patient-Friendly Injection Solutions: Prefilled Syringes, Cartridges & Pens
More -
2024-01-18
Bioworkshops has been selected as a "Unicorn" incubator in Suzhou in 2023
More






